United States Non-Invasive Prenatal Testing Market Size & Forecast (2025–2033)
Advancing Safer Prenatal Screening Through Innovation, Awareness, and Accessibility
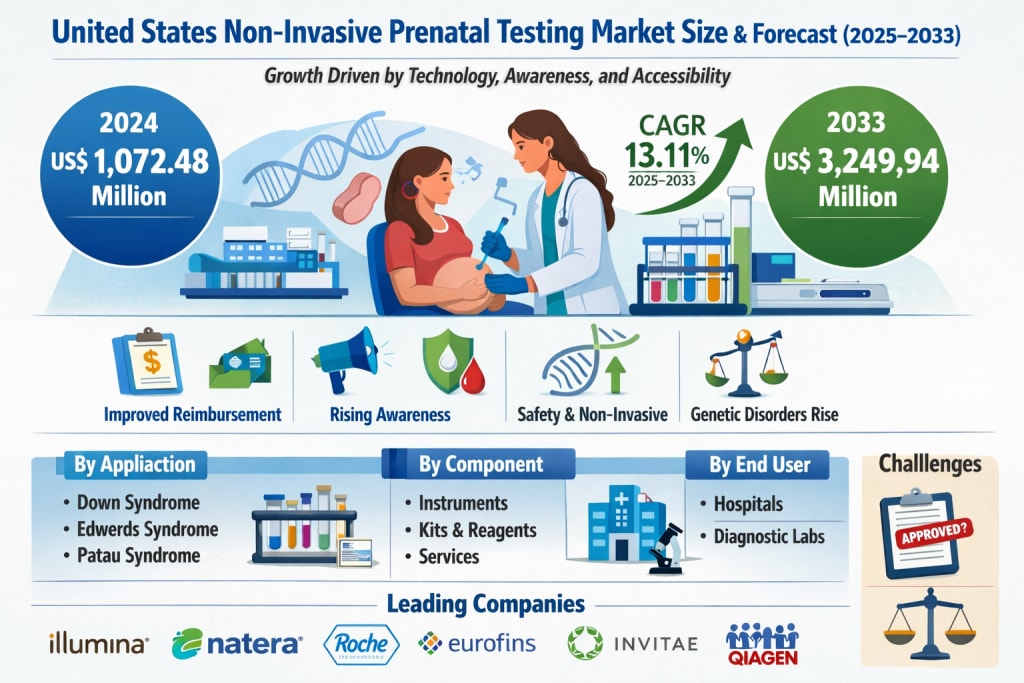
The United States Non-Invasive Prenatal Testing (NIPT) market is entering a transformative phase, fueled by technological breakthroughs, growing awareness among expectant parents, and increasing institutional support. According to Renub Research, the market is projected to grow from US$ 1,072.48 million in 2024 to US$ 3,249.94 million by 2033, expanding at a compound annual growth rate (CAGR) of 13.11% from 2025 to 2033.
This remarkable growth reflects a broader shift in prenatal care—from invasive, risk-prone diagnostic methods to safer, highly accurate screening technologies. NIPT, which analyzes fetal DNA circulating in a mother’s bloodstream, is now widely recognized as a reliable first-line screening option for detecting chromosomal abnormalities early in pregnancy.
Market Overview: A Shift Toward Safer and Smarter Screening
The rapid expansion of the NIPT market in the United States is largely attributed to advancements in genomic technologies and the increasing acceptance of non-invasive diagnostic approaches. Unlike traditional procedures such as amniocentesis or chorionic villus sampling (CVS), which carry a small but significant risk of miscarriage, NIPT offers a non-invasive alternative that requires only a maternal blood sample.
Through cutting-edge methods like next-generation sequencing (NGS), NIPT can detect conditions such as:
Down syndrome (Trisomy 21)
Edwards syndrome (Trisomy 18)
Patau syndrome (Trisomy 13)
Turner syndrome
In recent years, the scope of NIPT has expanded beyond common chromosomal abnormalities to include microdeletions and even certain single-gene disorders. This expansion has significantly enhanced its clinical utility and strengthened its role in modern prenatal care.
Another key driver is the endorsement of NIPT by leading medical bodies. Healthcare organizations now recommend it as a primary screening tool for all pregnant women, regardless of age or risk category. This universal recommendation marks a critical turning point, moving NIPT from a niche service for high-risk pregnancies to a mainstream healthcare solution.
Key Growth Drivers
1. Improved Reimbursement Policies
One of the most influential factors behind the market’s growth is the expansion of insurance coverage. Historically, the high cost of NIPT limited its accessibility, particularly for low- and middle-income populations. However, increasing inclusion of NIPT in both Medicaid and private insurance plans has significantly reduced out-of-pocket expenses.
This financial accessibility has:
Encouraged broader adoption across diverse demographics
Enabled early detection of genetic abnormalities
Improved overall prenatal care outcomes
As insurers continue to recognize the long-term cost benefits of early diagnosis—such as reduced complications and better care planning—coverage is expected to expand further, reinforcing market growth.
2. Rising Awareness and Clinical Recommendations
Awareness plays a crucial role in healthcare adoption, and NIPT is no exception. Over the past decade, educational campaigns, physician advocacy, and patient experiences have significantly increased awareness about the safety and accuracy of NIPT.
Professional guidelines now recommend NIPT as a first-line screening method for all pregnancies. This shift has led to:
Greater trust among patients
Increased physician adoption
Integration into routine prenatal care protocols
As more women become informed about the benefits of early, risk-free screening, the demand for NIPT continues to rise steadily.
3. Safety and Non-Invasiveness
Safety remains one of the strongest selling points of NIPT. Traditional diagnostic methods involve invasive procedures that can pose risks to both the mother and fetus. In contrast, NIPT eliminates these risks entirely.
Key advantages include:
No risk of miscarriage
Early testing (as early as 9–10 weeks of pregnancy)
High accuracy rates for common chromosomal conditions
This combination of safety and reliability makes NIPT particularly appealing to expectant parents who want reassurance without compromising fetal health.
4. Increasing Prevalence of Genetic Disorders
The rising incidence of chromosomal abnormalities is another significant driver. Down syndrome, for instance, remains one of the most common genetic conditions in the United States, with approximately 6,000 babies born each year, or about 1 in every 700 births.
This growing prevalence underscores the importance of early and accurate screening. As awareness of these conditions increases, so does the demand for reliable prenatal testing solutions like NIPT.
Market Challenges
1. Regulatory Oversight and Accuracy Concerns
Despite its advantages, the NIPT market faces notable regulatory challenges. Many NIPT tests are classified as laboratory-developed tests (LDTs), which are not subject to the same level of regulatory scrutiny as FDA-approved medical devices.
This lack of standardized oversight raises concerns about:
Variability in test accuracy
Risk of false positives or false negatives
Inconsistent quality across providers
False-positive results, in particular, can cause significant emotional distress for expectant parents and may lead to unnecessary follow-up procedures or difficult decisions.
Strengthening regulatory frameworks and establishing standardized guidelines will be critical to maintaining trust and ensuring consistent quality across the market.
2. Ethical and Social Implications
The growing use of NIPT also introduces complex ethical considerations. Early detection of genetic conditions can influence parental decisions regarding pregnancy continuation, raising sensitive questions about:
Selective termination
Genetic discrimination
Societal perceptions of disability
Additionally, interpreting genetic information can be challenging, often requiring counseling to help parents understand the implications of test results.
These ethical concerns highlight the need for:
Comprehensive genetic counseling services
Clear communication of test limitations
Responsible use of genetic information
Balancing technological advancement with ethical responsibility will be essential as the market continues to evolve.
Market Segmentation Analysis
By Component
Instruments: These include sequencing machines and diagnostic equipment used in laboratories. While essential, they represent a smaller share compared to consumables.
Kits and Reagents: This segment dominates the market due to recurring demand. Each test requires specialized reagents, making this a consistent revenue generator.
Services: Testing services offered by laboratories and diagnostic providers form a significant portion of the market, driven by increasing test volumes.
By Application
Down Syndrome (Trisomy 21): The largest segment, driven by high prevalence and strong clinical focus.
Edwards Syndrome (Trisomy 18) and Patau Syndrome (Trisomy 13): Though less common, these conditions are critical targets for early detection.
Turner Syndrome: Increasingly included in expanded screening panels.
Other Applications: Includes microdeletions and emerging genetic conditions, reflecting the expanding capabilities of NIPT technologies.
By End User
Hospitals: Major contributors due to integrated prenatal care services and higher patient volumes.
Diagnostic Laboratories: Play a crucial role in processing tests and delivering results, often partnering with healthcare providers.
Industry Developments and Innovation
The NIPT market is highly dynamic, with continuous innovation shaping its trajectory. In May 2024, a major advancement was introduced with the launch of a cfDNA-based fetal RhD test, enabling early detection of fetal RhD status using maternal blood samples.
This development is particularly significant given the shortage of Rho(D) immune globulin (RhIg) treatments in the United States. By identifying RhD status as early as nine weeks into pregnancy, healthcare providers can make more informed treatment decisions, improving patient outcomes.
Such innovations highlight the expanding scope of NIPT beyond chromosomal screening, positioning it as a comprehensive tool in prenatal diagnostics.
Competitive Landscape
The United States NIPT market is characterized by intense competition among leading biotechnology and diagnostics companies. Key players are focusing on:
Expanding test portfolios
Enhancing accuracy and reliability
Investing in research and development
Forming strategic partnerships
Major companies operating in the market include:
Eurofins Scientific
F. Hoffmann-La Roche Ltd
Invitae Corporation
Illumina Inc.
Natera Inc.
Centogene NV
Qiagen
These organizations are leveraging advanced genomic technologies and data analytics to maintain competitive advantage and meet evolving market demands.
Future Outlook: A Market Poised for Continued Growth
The future of the U.S. NIPT market looks exceptionally promising. Several trends are expected to shape its evolution over the next decade:
Expansion into broader genetic screening: Including rare diseases and personalized medicine applications
Integration with digital health platforms: Enhancing data interpretation and patient engagement
Global standardization of testing protocols: Improving consistency and reliability
Increased focus on ethical frameworks: Ensuring responsible use of genetic information
As technology continues to advance and healthcare systems prioritize early diagnosis and preventive care, NIPT is set to become an integral part of prenatal healthcare worldwide.
Final Thoughts
The United States Non-Invasive Prenatal Testing market represents a powerful intersection of innovation, accessibility, and patient-centered care. With its ability to provide early, accurate, and risk-free insights into fetal health, NIPT is redefining the standards of prenatal screening.
While challenges related to regulation and ethics remain, the overall trajectory of the market is undeniably positive. Backed by strong growth projections, expanding applications, and increasing acceptance, NIPT is not just a technological advancement—it is a transformative force in modern healthcare.






Comments
There are no comments for this story
Be the first to respond and start the conversation.